How To Solve Mole Fraction Problems
And finally we can determine the mass of BDA and water in the gas in the room. For a mixture of two substances A and B the mole fractions of each would be written as follows.

What Are The Mole Fractions Of H3po4 And W Clutch Prep
M of H 2 O is 18 and M of CH 3 OH is 32.
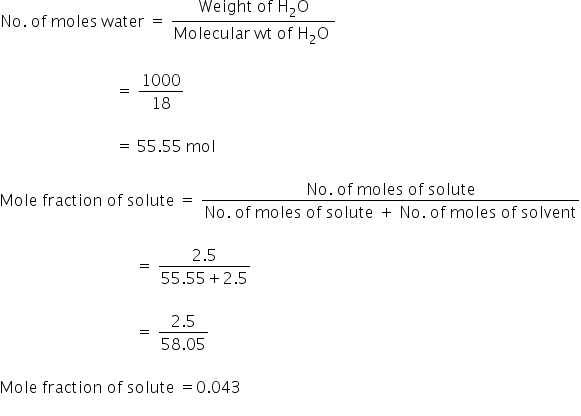
How to solve mole fraction problems. An atom of some element X weighs 6644 10 -23 g. The most common technique is finding the total number of moles of solute dissolved. Mole fraction of CH 3 OH 017 22 017.
Mole Fraction Problems Category. C 12 H 22 O 11. Mole fraction is a unitless and dimensionless expression.
Advantages of molar fraction. Multiplying the mole fraction by 100 gives the mole percentage also referred as amountamount percent abbreviated as nn. Moles of CH 3 OH 55 32 017 mole.
Mass number of moles molar mass where mass is in grams and the molar mass is in grams per mole. Then by assuming the gas phase is an ideal gas we can determine the total mass of air in the room. In the given mixture sum of all the mole fractions is equal to one.
Mass of 1 mole X atoms. We can easily convert mole percent back to mole fraction by dividing by 100. Let xC12H22O11 and xH2O be the mole fraction of sucrose and water in the solution.
C 12 H 22 O 11 12 12 1 22 16 11 C 12 H 22 O 11 144 22 176. Chemical Engineering Math Published in Newark California USA Calculate the mole fractions of ethyl alcohol C2H5OH and water in a solution made by dissolving 92 grams of alcohol in 18 grams of H2O. We can convert the mole fraction into a mass fraction.
Solute is 4 g of C 12 H 22 O 11. Step 1 - Determine number of moles of sucrose in 4 g. Mole fraction of CH 3 OH 0073.
So the atomic mass of X 40. If a mixture consists of 050 mol A and 100 mol B then the mole fraction of A would be X A 05 15 033. Therefore according to the equation.
Thus the solution has 0021 of sugar and 0979 of water. 6644 10 -23 602210 23. Determine the mole fraction of CH 3 OH and H 2 O in a solution prepared by dissolving 55 g of alcohol in 40 g of H 2 O.
Calculate the number of gram-atoms in 40 kg of it. 14121 X A mol A mol A mol B and X B mol B mol A mol B. For general chemistry all the mole percents of a mixture add up to 100 mole percent.
X A X B 1. If there is one solution having very low in concentration then it is called as the dilute solution and a solution with higher energy or concentration is called as the concentrated solution. Of gram-atoms or moles of X.
So it would not vary from sample to sample. Mole fraction is not dependent on the temperature. Molality is the number of moles of solute per kilogram of solvent.
Moles to Mass Calculation We can use the above equation to find the mass of a substance when we are given the number of moles of the substance. Moles of H 2 O 40 18 22 moles. The multiplication of the mole fraction by 100 gives the mole percentage.
The mole fraction is an intensive property. Mass of 1 atom Avogadro constant.

Aleks Calculating Mole Fraction In A Gas Mixture Youtube
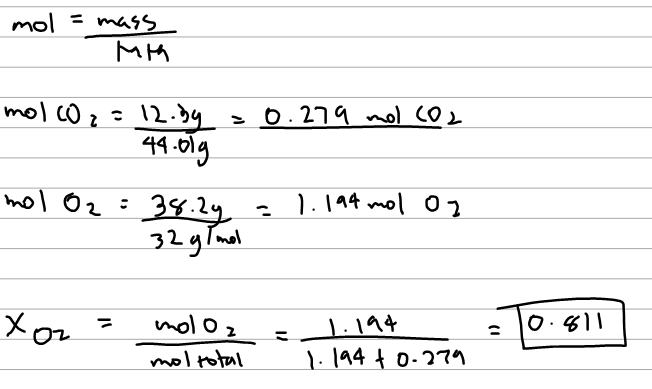
What Is The Mole Fraction Of O2 In This Mi Clutch Prep

Calculate The Molality Molarity And Mole Clutch Prep
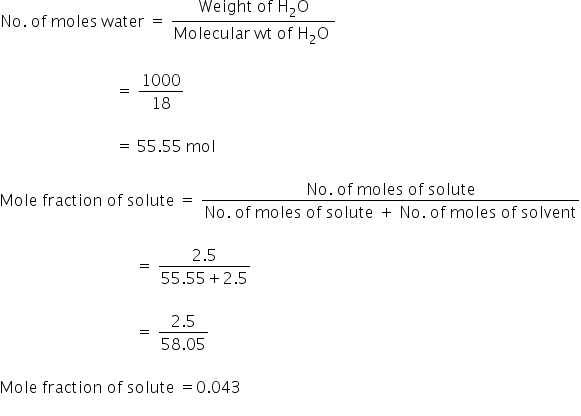
What Is A Mole Fraction Of A Solute In 225m Aqueous Solution Chemistry Topperlearning Com G0zrdree

Mole Fraction Definition Formula Symbol Examples Chemistrygod
Mole Fraction Definition Formula Symbol Examples Chemistrygod

15 4c Calculating Mole Fraction Youtube

What Is The Mole Fraction Of Oxygen Gas In Clutch Prep

Chemistry Solutions 12 Of 53 Expressing Concentrations Mole Fraction Example 2 Youtube
Mole Fraction Definition Formula Symbol Examples Chemistrygod
How To Relate Mole Fraction And Molality Quora

How To Calculate Mole Fraction Examples Explanation Shortcut Equation Practice Problems Youtube

Calculate Molality Molarity And Mole Fraction Short Cuts

Mole Fraction Class Xi Freeguru Helpline
Molarity And Solution Units Of Concentration
What Is The Mole Fraction Of A Solute Quora




